Porcine Bone Graft
By: Citagenix
Last Updated: 10/12/2024
$69.98
THE Graft is a natural, porous bone mineral matrix produced by removal of all organic components from porcine cancellous bone. Due to its natural structure, the anorganic bone mineral of THE Graft TM mimics the physical and chemical aspects of mineralized matrix of human bone. When packed into a bone defect, THE Graft TM gradually resorbs and is replaced with new bone during the healing process.
- Safe & Biocompatible. Higly Level of Purity.
- Backed by Extensive Research
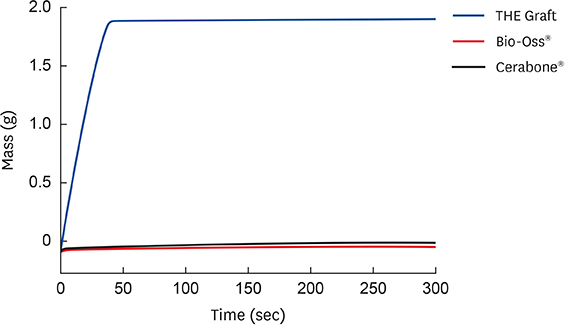
- Higher Hydrophilicity and Higher Porosity than comparable xenografts
- Optimal Osteoconductivity
- Available in Vials .25-1mm particle and various sizes. Choose below.
- Compare to BioOss ®1 (Note: Source of BioOss is bovine. Source of The Graft is porcine.)
You May Also Like:
Additional Information for Porcine Bone Graft
THE Graft TM is designed to mimic the structure of human bone, offering osteoconductive properties to host cell growth and complete remodeling to host bone. The natural structure of the anorganic bone mineral of THE Graft demonstrates similar physical and chemical characteristics compared to the mineralized matrix of human bone. THE Graft gradually resorbs and is replaced with host bone during the healing process. THE Graft is cancellous granules and is sterilized using gamma irradiation.
THE Graft TM Features & Benefits
- THE Graft TMBiocomptability: Getting closer to human bone
The Graft is structurally similar to human tissue. It has high possible level of porosity combined with a natural interconnectivity. This supports absorption of liquids such as blood and leads to incorporation and remodeling. In addition, the combination of porcine origin with the high level of purity enables predictable bone growth without risking an immunogenic reaction. The high biocompatibility of THE Graft™ has been confirmed by an in-vitro cell study. THE Graft™ therefore encourages cell adhesion to the same extent as the established natural DBBM and offers optimal conditions for vital cell growth. - THE Graft TM Purity and Safety
Patented proprietary virus inactivation process technology. Thanks to highly efficient patented manufacturing process, THE Graft™ is free from any organic components that might be potential causes of infection or immune reaction. This unique process preserves most of the physical properties of the native porcine osseous structure of THE Graft™. A large surface area is a key requirement for graft materials, and not only results in a larger surface region available for osteoblast cells attachment but also facilitates the exchange of nutrients and waste products, it allows greater amounts of blood, proteins, and growth factors to be absorbed onto the scaffold. - THE Graft™ Hydrophilicity and Optimal Osteoconductivity
THE Graft™ consists of a unique inter-connection pore system that ensures an efficient fluid intake and permits the migration of cells. This pore system and high surface energy enhances the osteoconduction process. The wettability of THE Graft™ turned out to be higher than compared existing xenografts, which suggests that THE Graft™ is relatively hydrophilic and can be easily wet by body fluids after implantation. Not only protein adsorption, but also the attachment, growth, and proliferation of various types of cells, including osteoblasts, have been reported to be significantly affected by the wettability of the material surface. - The Graft: High Porosity
The High porosity of THE Graft™ means a quicker absorption of liquides (e.g;blood) in comparision with DBBM. This not only facilitates the application of the material but also leads to a quicker post-implantation incorporation. High level of porosity was demonstrated with particle pore structure test, particle size distribution test and total porosity tests. - Minimal residual protein without the use of high temperature sintering. The natural interconnectivity is maintained. THE Graft TM is structurally similar to human bone.
THE Graft TM Clinical References
1. J Periodontal Implant Sci. 2017 Dec;47(6):388-401. Physicochemical characterization of porcine bone-derived grafting material and comparison with bovine xenografts for dental applications.
Considering that both bovine bone grafting materials have been successfully used in oral surgery applications in the last few decades, this work shows that the porcine-derived grafting material possesses most of the key physiochemical characteristics required for its application as a highly efficient xenograft material for bone replacement.
2. Ceramics International Volume 36, Issue 8, December 2010, Pages 2383-2393 Effect of the calcination temperature on the composition and microstructure of hydroxyapatite derived from human and animal bone, M. Figueiredo, A, Fernando, G. Martins, J. Freitas, F.Judas, H, Figueiredo
3. J Biomed Mater Res A. 2014 Oct;102(10):3609-17. doi: 10.1002/jbm.a.35022. Epub 2013 Nov 18. Osteogenic effect of low-temperature-heated porcine bone particles in a rat calvarial defect model., Go A1, Kim SE, Shim KM, Lee SM, Choi SH, Son JS, Kang SS.
In this study, low crystalline bone particles were obtained at low processing temperature (at temperature of 400°C) and achieved superior new bone formation compared with the high crystalline bone particles created at a higher process temperature (1200°C). It can be concluded that lower process temperature bone particles might provide a more effective graft material for enhancing bone formation.
4. Clin Implant Dent Relat Res 2018 Jul 27. Epub 2018 Jul 27. Alveolar ridge regeneration of damaged extraction sockets using deproteinized porcine versus bovine bone minerals: A randomized clinical trial. 100 patients
It is therefore suggested that DBBM (deproteinized bovine) and DPBM (deproteinized porcine) can be similarly used for grafting in damaged extraction sockets that lack bony walls.
5. Clin Implant Dent Relat Res. 2017; 19(1):140-150 Randomized Clinical Trial of Maxillary Sinus Grafting using Deproteinized Porcine and Bovine Bone Mineral. 16 Patients
The results suggested that DPBM might produce comparable bone formation and volumetric stability with DBBM in maxillary sinus grafting, however, further clinical study with longerâÂÂterm periods and larger sample sizes should be needed for confirming this suggestion.
6. Journal of Microbiology 52.2 (2016): 140-147. “Process development of a virally-safe dental xenograft material from porcine bones.” The Korean Journal of Microbiology 52.2 (2016): 140-147.
T